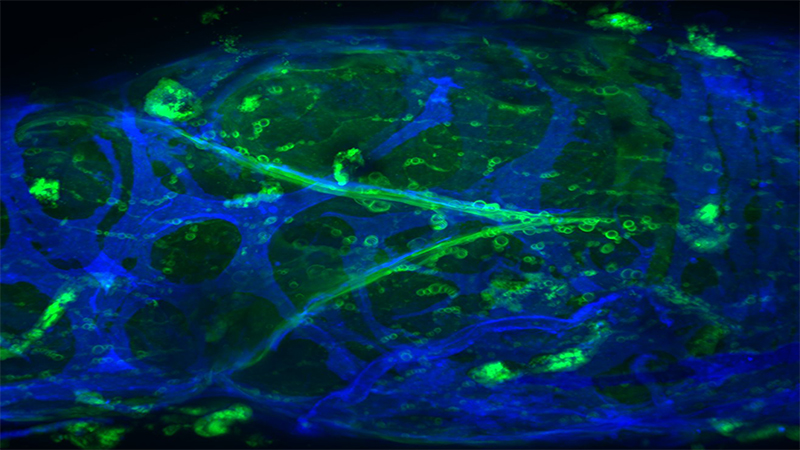
Imagine dissecting something so small as a mouse’s lymphatic vessel, a tube half the diameter of a human hair. Now, imagine inserting an even tinier pipette into that vessel — a procedure called cannulation that requires a steady index finger and a microscope that magnifies an object to 90 times its actual size. To top it off, imagine doing this after downing a double espresso.

You’ve imagined a typical morning for Michael Davis, PhD.
“The number of people in the world who can do these physically fine micro-dissections, you could probably count on two hands,” said Scott Zawieja, PhD, who was a postdoctoral fellow in Davis’ lab before recently accepting a position at MU as an assistant professor of medical pharmacology and physiology. “The number of people who have the dedication to do it day in and day out, even on the weekends, it dwindles quite a bit more.”
And the number of people who can cannulate a mouse lymphatic vessel while under the influence of an infusion of concentrated coffee?
That’s a party of one.
“Now that I’m older, I guess I’m less sensitive to caffeine,” Davis explained.
Davis, a Curators' Distinguished Professor and the Margaret Proctor Mulligan Professor of Medical Research, is an international expert on lymphatics, the vascular system that moves fluid containing white blood cells through the body to fight disease and remove waste. He recently received a continuation of his National Institutes of Health R01 grant that is worth $2.54 million over the next four years to study lymphatic pace-making.
His ultimate goal is to contribute to a treatment for lymphedema, a condition marked by extreme swelling, usually of an arm or leg, that develops when lymphatic vessels quit pumping fluid. It often affects patients whose lymph nodes have been removed or damaged during cancer treatments.
“When I finish, we’ll have a very good idea of the specific targets in lymphatic muscle that we can manipulate pharmacologically,” he said.
Once that is determined, a treatment could be developed for patients with lymphedema to target those proteins and improve the function of the vessels so they begin pumping fluid again.
A new method
Davis’ research is an example of the work the University of Missouri is encouraging with the NextGen Precision Health Initiative. Davis said about 300 scientists worldwide are dedicated to studying the lymphatic system, but the only ones conducting groundbreaking experiments on the dissected lymphatic vessels of mice are Davis and the people he has trained. That makes him a highly sought collaborator and makes his lab a beacon for young scientists interested in lymphatics.
“He’s the reason I came here,” said Zawieja, who has received his own NIH grant to study lymphatics. “Since the late 2000s, he’s pioneered a new method of lymphatic research, using next-generation technologies in mouse transgenics.”
That method comes with a steep learning curve. Jorge Castorena-Gonzalez, PhD, who recently completed his postdoctoral fellowship in Davis’ lab, said the first time he tried to dissect one of the nearly translucent lymphatic vessels, he couldn’t see what he was supposed to be removing. Even when he managed to dissect a vessel, he couldn’t insert the glass pipette without breaking it. The pipettes are so small, Davis has to make them himself with a machine he jury-rigged for the job, so every time Castorena-Gonzalez broke a pipette, he had to make the walk of shame to his boss’ office and ask him to create a new one.
This went on eight hours a day for two months. Castorena-Gonzalez recalled trudging back to his car at night, thinking he was letting Davis down and that he would never master the technique. He finally did, and now he has his own NIH grant and a job as an assistant professor of pharmacology at Tulane University. “He’s very patient with everyone he trains,” Castorena-Gonzalez said. “I can only imagine it has to get a little frustrating when you spend so much time training someone and that person keeps failing and failing and failing. But if he’s frustrated, he never shows it. He’s always very calm.”
Davis developed an interest in the lymphatic system by accident. He was studying blood vessels at Texas A&M University when, one day, a colleague from across the hall popped in and asked if he had any ideas on how to cannulate a rat’s lymphatic vessel. It was practically a firehose compared to the mice vessels Davis now works on, but it was quite a challenge at the time. Davis created devices to steady the vessel, hold the pipette and anchor the user’s hand.
The project piqued his interest, and for the last 30 years, he’s been absorbed with figuring out why lymphatic vessels work and why they fail. Aside from daily swims and the occasional round of golf, his only hobby is creating contraptions and techniques for extracting information from those tiny tubes. “He’s a constant tinkerer,” Zawieja said. “If you come in here on the weekends, you’ll see him working on the next new piece of technology that can get him some answers that nobody else is willing to put the time or effort into understanding.”
Closer to a cure
In his previous research, Davis made a breakthrough discovery about the chemical process behind lymphatic pumping. Unlike the vascular system, in which the heart pumps blood, there is no pump that powers the lymphatic system. The vessels themselves contract and relax to move fluid. He studied 15 different ion channels — they are the gatekeepers on a cell membrane that determine if electrically charged molecules are allowed to pass in or out of the cell — and found only one channel affected contractions. When fluid builds up in tissue, the pressure causes a change in calcium levels, triggering a chloride ion channel to open up and release chloride ions. The release of those ions causes the vessel to contract and pump fluid.
Davis determined this by applying different levels of pressure to the vessels of 15 types of genetically altered mice. The vessels of mice that lacked the chloride ion channel were the only ones that didn’t respond to the changes in pressure.
With his new grant, Davis is looking deeper into the process to examine which GPCRs — G protein coupled receptors — are signaling the chloride ion channel to open when the calcium level changes. He will try to determine if the failure of those GPCRs is causing the communication breakdown that prevents the vessels from contracting. That discovery could ultimately lead to a lymphedema drug that targets the appropriate GPCRs.
“We would be able to go back and intervene there and rescue contraction in a vessel that has stopped contracting,” Davis said.
He isn’t sure if all his work will lead to a cure for lymphedema before he packs up the microscope and espresso machine, but he is confident the scientists he’s trained at MU will carry on his work and take it in new directions, albeit without so much caffeine in their bloodstream.

Highlighting the promise of personalized health care and the impact of large-scale interdisciplinary collaboration, the NextGen Precision Health initiative is bringing together innovators from across the University of Missouri and the UM System’s three other research universities in pursuit of life-changing precision health advancements. It’s a collaborative effort to leverage the research strengths of Mizzou toward a better future for the health of Missourians and beyond. The Roy Blunt NextGen Precision Health building at MU anchors the overall initiative and expands collaboration between researchers, clinicians and industry partners in the state-of-the-art research facility.